Abstract
Research Article
Audit of the effect of non-nasal specific scoring on the Postoperative SNOT22 questionnaire
Samir Gendy*
Published: 23 September, 2020 | Volume 4 - Issue 1 | Pages: 003-005
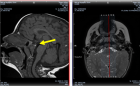
The Sino-nasal outcome test (SNOT22) has been widely adopted in clinical practice and has been declared as the most suitable sinonasal outcome scoring system. It is simple disease specific encompassing 22 symptoms reflecting health burden of the rhino- logical patients.
Each item quantifies symptoms severity from 0(no problem) to 5 (worst symptom). The sum of each item results in a maximum score of 110. High score indicates poor outcome.
Read Full Article HTML DOI: 10.29328/journal.ated.1001010 Cite this Article Read Full Article PDF
References
- Snidvongs K, Heller GZ, Harvey RJ. Validity of European Position Paper on Rhino sinusitis disease control assessment and modification in Chronic Rhino sinusitis.Otolaryng. Head Neck Surg. 2014; 150: 479-486. PubMed: https://pubmed.ncbi.nlm.nih.gov/24343022/
- Kordjian HH. Sixty-Three patient-based survey-Can SNOT-22 test be a suitable evaluation method for septoplasty and turbinectomy Clin.Otolaryng. 2017; 42: 1373-1377
- Zachary M, Jones R, le P, Rudmik L, Mattos JL, et al. SNOT-22 outcomes after sinus surgery a systematic review and meta-analysis. Laryngoscope. 2018; 128: 581-592. PubMed: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5814358/
- Kennedy J, Hubbard M, Huyett P, Patrie JT, Borish L, et al. SNOT-22: A predictor of post-surgical improvement in patients with chronic sinusitis. Ann Allergy Asthma Immunol.2013; 111: 246-251. PubMed: https://pubmed.ncbi.nlm.nih.gov/24054358/
Figures:

Figure 1
Similar Articles
-
A possible Etiology and new treatment of Burning Mouth Syndrome and allied conditionSudhakar Pattanaik*. A possible Etiology and new treatment of Burning Mouth Syndrome and allied condition. . 2017 doi: 10.29328/journal.ated.1001001; 1: 001-005
-
Changes in the frequency and intensity of Tinnitus using the Suppressive Noise SpectrumMing Zhang*,Alysia Jeske,Sarah Young. Changes in the frequency and intensity of Tinnitus using the Suppressive Noise Spectrum. . 2017 doi: 10.29328/journal.ated.1001002; 1: 006-012
-
Practical implementation of the SWEEP-session of Stimulation-Registration in CI fittingPetrov SM*. Practical implementation of the SWEEP-session of Stimulation-Registration in CI fitting. . 2017 doi: 10.29328/journal.ated.1001003; 1: 013-015
-
For professionals working on the topic of cochlear implantation: Opinions of readers of “Instruction” and participants of MIMICPetrov SM*. For professionals working on the topic of cochlear implantation: Opinions of readers of “Instruction” and participants of MIMIC. . 2018 doi: 10.29328/journal.ated.1001004; 2: 001-005
-
Recent advances in pathophysiology and management of subglottic HemangiomaMohamed Khamis Tolba Mahmoud Abdalla*. Recent advances in pathophysiology and management of subglottic Hemangioma. . 2018 doi: 10.29328/journal.ated.1001005; 2: 006-007
-
Topical Management of chronic rhinosinusitis - A literature reviewAremu Shuaib Kayode*,Tesleem Olayinka Orewole. Topical Management of chronic rhinosinusitis - A literature review. . 2019 doi: 10.29328/journal.ated.1001006; 3: 001-006
-
Feasibility study on the evaluation of the effect of narrow-band CE-Chirp ASSR in the hearing field after hearing aid in hearing-impaired childrenWang Yonghua*,Xing Shuoyao. Feasibility study on the evaluation of the effect of narrow-band CE-Chirp ASSR in the hearing field after hearing aid in hearing-impaired children. . 2019 doi: 10.29328/journal.ated.1001007; 3: 007-011
-
Prevalence of disabling hearing loss in the elderlyLetícia Raquel Baraky Vasconcelos,Paula Ferraz Rodrigues,Paula Liziero Tavares,Audryo Oliveira Nogueira*. Prevalence of disabling hearing loss in the elderly. . 2019 doi: 10.29328/journal.ated.1001008; 3: 012-013
-
Impact of Community Oriented Ear Care (COEC) on national programme for control of deafness in India: A critical lookSanjeev Davey*,Anuradha Davey,Rajesh Jain. Impact of Community Oriented Ear Care (COEC) on national programme for control of deafness in India: A critical look. . 2020 doi: 10.29328/journal.ated.1001009; 4: 001-002
-
Audit of the effect of non-nasal specific scoring on the Postoperative SNOT22 questionnaireSamir Gendy*. Audit of the effect of non-nasal specific scoring on the Postoperative SNOT22 questionnaire. . 2020 doi: 10.29328/journal.ated.1001010; 4: 003-005
Recently Viewed
-
Looking Beyond Common Causes of Renal Dysfunction: Renal GVHD and Thrombotic Microangiopathy after Allogeneic TransplantTaner Tan*,Ahmet Umur Topcu,Erdem Cig,Dilek Ertoy Baydar,Sinem Civriz Bozdag. Looking Beyond Common Causes of Renal Dysfunction: Renal GVHD and Thrombotic Microangiopathy after Allogeneic Transplant. Insights Clin Cell Immunol. 2026: doi: ; 10: 001-003
-
Screening for Depressive Symptoms in Clinical and Nonclinical Youth: The Psychometric Properties of the Dutch Children’s Depression Inventory-2 (CDI-2)Denise HM Bodden*,Yvonne Stikkelbroek,Daan Creemers,Sanne PA Rasing,Elien De Caluwe,Caroline Braet. Screening for Depressive Symptoms in Clinical and Nonclinical Youth: The Psychometric Properties of the Dutch Children’s Depression Inventory-2 (CDI-2). Insights Depress Anxiety. 2025: doi: 10.29328/journal.ida.1001047; 9: 028-039
-
From the Single Bacterial Cell to the Microbial Community: A Round Trip to better understand the Secrets of Complex Microbiological EcosystemsErasmo Neviani*. From the Single Bacterial Cell to the Microbial Community: A Round Trip to better understand the Secrets of Complex Microbiological Ecosystems. Int J Clin Microbiol Biochem Technol. 2024: doi: 10.29328/journal.ijcmbt.1001029; 7: 006-008
-
Renal Malakoplakia: A Diagnostic Challenge Presenting as a Subcapsular Collection despite Clinical RecoveryMohammed Amine Elafari*,Mamad Ayoub,Mohammed Amine Bibat,Amine Slaoui,Tarik Karmouni,Abdelatif Koutani,Khalid Elkhader. Renal Malakoplakia: A Diagnostic Challenge Presenting as a Subcapsular Collection despite Clinical Recovery. J Clin Med Exp Images. 2026: doi: 10.29328/journal.jcmei.1001046; 10: 032-038
-
Not Every Bladder Mass Is Malignant: A Case of Inverted Urothelial Papilloma in a Young AdultMohammed Amine Elafari*,Mamad Ayoub,Mohammed Amine Bibat,Amine Slaoui,Tarik Karmouni,Abdelatif Koutani,Khalid Elkhader. Not Every Bladder Mass Is Malignant: A Case of Inverted Urothelial Papilloma in a Young Adult. J Clin Med Exp Images. 2026: doi: 10.29328/journal.jcmei.1001043; 10: 016-019
Most Viewed
-
Physical Performance in the Overweight/Obesity Children Evaluation and RehabilitationCristina Popescu, Mircea-Sebastian Șerbănescu, Gigi Calin*, Magdalena Rodica Trăistaru. Physical Performance in the Overweight/Obesity Children Evaluation and Rehabilitation. Ann Clin Endocrinol Metabol. 2024 doi: 10.29328/journal.acem.1001030; 8: 004-012
-
Hypercalcaemic Crisis Associated with Hyperthyroidism: A Rare and Challenging PresentationKarthik Baburaj*, Priya Thottiyil Nair, Abeed Hussain, Vimal MV. Hypercalcaemic Crisis Associated with Hyperthyroidism: A Rare and Challenging Presentation. Ann Clin Endocrinol Metabol. 2024 doi: 10.29328/journal.acem.1001029; 8: 001-003
-
Effects of dietary supplementation on progression to type 2 diabetes in subjects with prediabetes: a single center randomized double-blind placebo-controlled trialSathit Niramitmahapanya*,Preeyapat Chattieng,Tiersidh Nasomphan,Korbtham Sathirakul. Effects of dietary supplementation on progression to type 2 diabetes in subjects with prediabetes: a single center randomized double-blind placebo-controlled trial. Ann Clin Endocrinol Metabol. 2023 doi: 10.29328/journal.acem.1001026; 7: 00-007
-
Exceptional cancer responders: A zone-to-goDaniel Gandia,Cecilia Suárez*. Exceptional cancer responders: A zone-to-go. Arch Cancer Sci Ther. 2023 doi: 10.29328/journal.acst.1001033; 7: 001-002
-
Ectopic adrenal tissue at the spermatic cordAbdallah Chaachou,Nizar Cherni,Wael Ferjaoui*,Mohamed Dridi,Samir Ghozzi. Ectopic adrenal tissue at the spermatic cord. J Clin Med Exp Images. 2022 doi: 10.29328/journal.jcmei.1001024; 6: 001-002

If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."